MEDtube is the largest social eLearning platform for 350,000+ professionals sharing 30,000+ videos, courses, images, documents and webinars
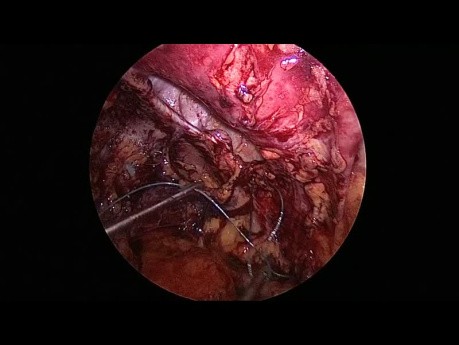
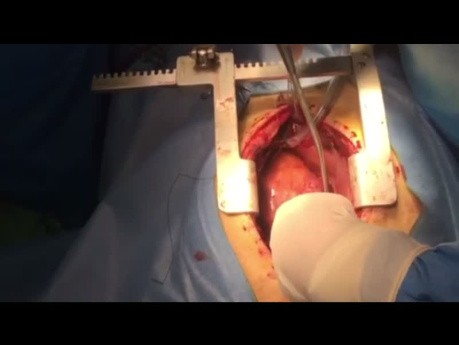
Register for freeLaparoscopic Transabdominal Preperitoneal Left...
This video depicts the laparoscopic repair of an L4, left subcostal incisional hernia via a transabdominal preperitoneal approach.
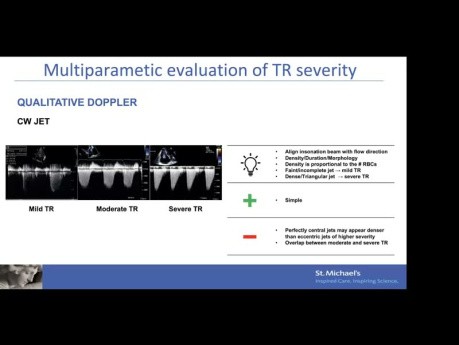
Echo Assessment of Tricuspid Regurgitation –...
Echo assessment of tricuspid regurgitation – mechanism, quantification and device selection. Speaker: Mathias Claeys MD PhD, Interventional Echocardiography Fellow, St. Michael's Hospital. Objectives:...
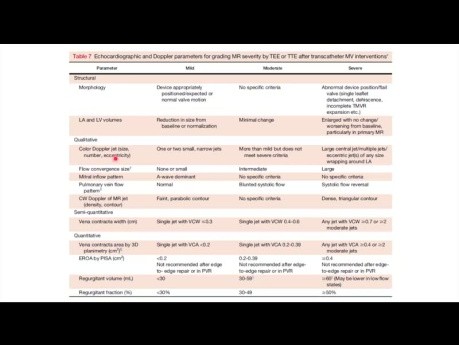
Case Potpourri: Regurgitation after Transcatheter...
Regurgitation after transcatheter edge-to-edge repair. Date: Thursday, 1 Feb 2024 Speaker: Flora Huang, MD FRCPC Echo Fellows, St. Michael’s Hospital. Objectives: 1. Review assessment of valvular...
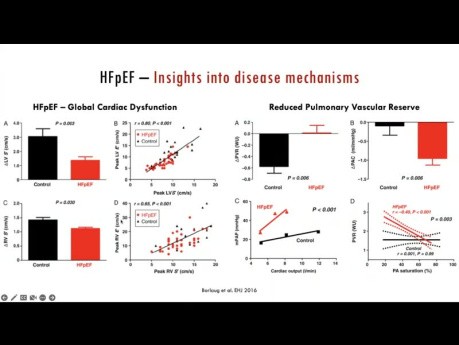
Stressing the Stepchild - Clinical Utility of...
Stressing the stepchild - Clinical Utility of Evaluating the RV-PV Unit During Exercise. Speaker: Mathias Claeys, MD PhD. Interventional Echocardiography Fellow, St. Michael's Hospital. Twitter:...
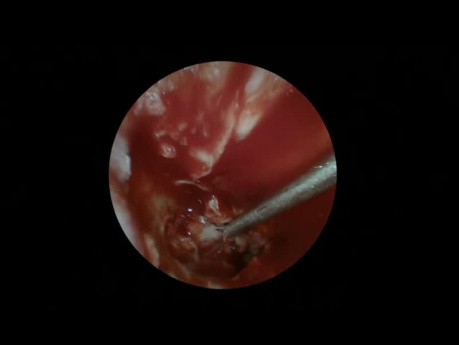
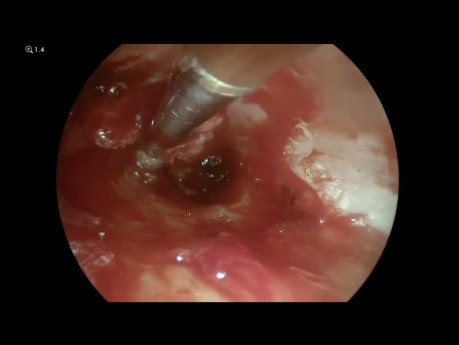
Chronic Suppurative Otitis Media Tympanoplasty
ENT video case: this is a demonstration of an endoscopic tympanoplasty as a treatment for chronic suppurative otitis media.
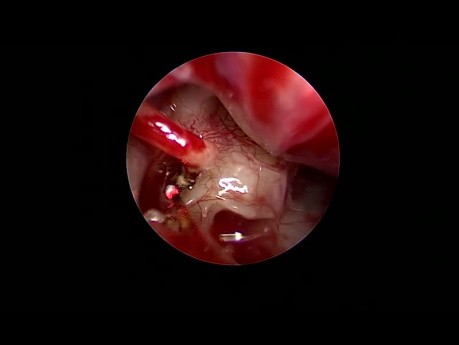
Endoscopic Stapedotomy for Otosclerosis (Diode...
We decided to try an old diode laser we had on the OR. It has a long fibre that is adjustable for any need. We used it to cut the stapes tendon, the posterior crus of the stapes suprastructure and for...
Shoulder Replacement - What You Need to Know
In this comprehensive video, we delve into the world of shoulder replacement surgery. Whether you're a patient seeking more information or a healthcare professional aiming to expand your knowledge, this...
Simultaneous Heart and Kidney Transplant
Simultaneous heart and kidney transplant, also known as a dual organ transplant, is a complex surgical procedure that involves the transplantation of both a heart and a kidney from a deceased donor into...
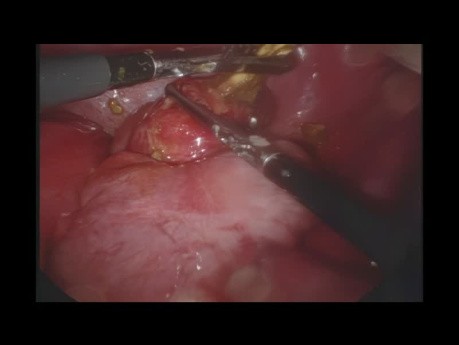
Laparoscopic Removal of a Perforated Foreign...
This video shows a case with laparoscopic treatment of a foreign body which has perforated the cecum.
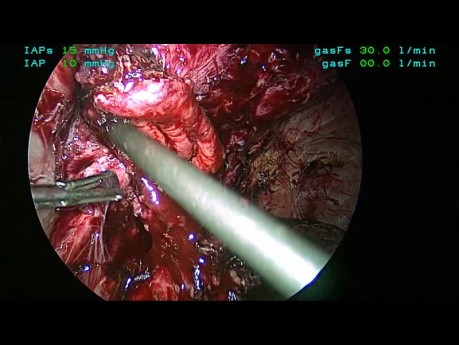
Laparoscopic Removal Of Eroded Mesh And Revision...
This video depicts the laparoscopic removal of mesh which has eroded into the gastrojejunostomy. The mesh was placed at the time of a prior laparoscopic repair of hiatal hernia and Roux-en-Y gastric bypass.
Featured channels
Mitral Valve Repair Center at The Mount...
As one of the world's most respected and experienced heart…
EUS - ENDO
EUS-ENDO will offer one day of live transmission from Paoli-Calmettes…
SAGES - Society of American Gastrointestinal...
The Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) is…