MEDtube is the largest social eLearning platform for 350,000+ professionals sharing 30,000+ videos, courses, images, documents and webinars
Register for freeProximal Humerus Radiographic Parameters
Orthopaedic video: proximal humerus radiographic parameters. Join the channel membership to unlock access to premium courses.
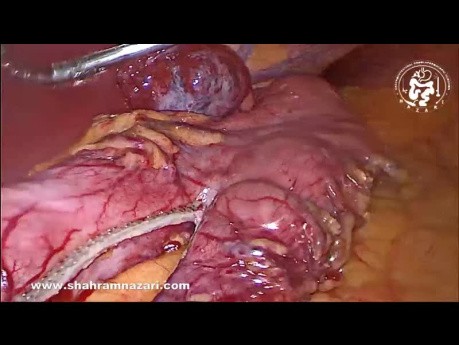
Giant Liver Hemangioma as an Incidentaloma in...
Different laparoscopic bariatric surgeries including laparoscopic sleeve gastrectomy (LSG), Roux-en-Y gastric bypass surgery (RYGB), and adjustable gastric banding (AGB) have become increasingly popular...
Using Artificial Intelligence in The Prediction...
Using Artificial Intelligence in The prediction of CAD with FFR and CT FFR or simple CTA.
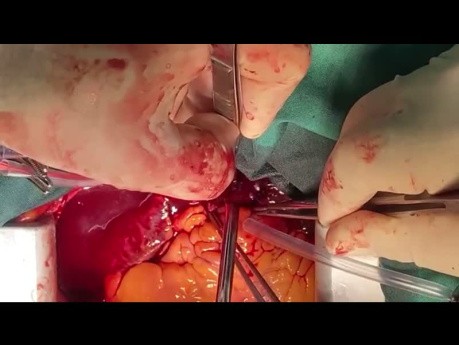
Off Pump Pulmonary Embolectomy a New Paradigm...
Pulmonary embolectomy is a surgical procedure performed to remove blood clots, known as emboli, from the pulmonary arteries in the lungs. These clots typically originate from deep vein thrombosis (DVT)...
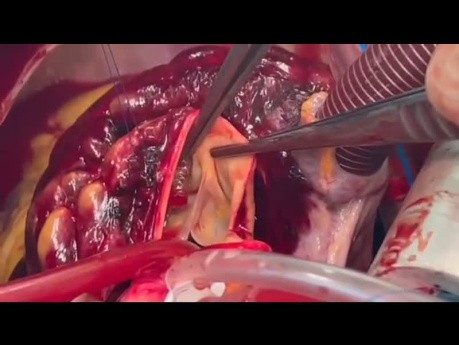
Stent Fracture of LT Main Ostia
Surgical video case: coronary artery stent fracture is a complication that can occur after the placement of a stent in a coronary artery to treat coronary artery disease. Stent fracture involves a break...
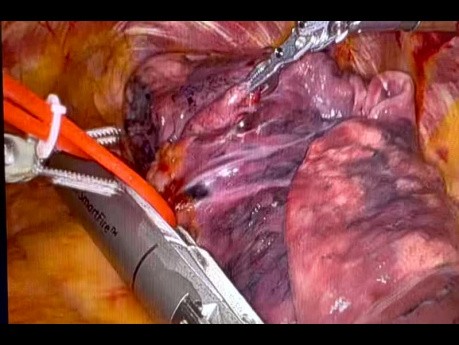
Robotic Stapler For Left Upper Lobectomy
Robotic stapler for left upper lobectomy. Tessitore A, Patella M, Casiraghi M and Cafarotti S. Ente Ospedaliero Cantonale-Ticino, Switzerland.
Versius Robotic Middle Lobectomy
This is a great video of a Cambridge Medical Robotics Pulmonary Lobectomy. This is the middle lobe used in conjunction with the new 8mm vascular stapler.
Complex Cataract Surgery with Missing Zonule...
This video case demonstrates a complex cataract with surprise of missing zonules found during the case. Techniques to avoid complications and achieve a stable IOL implant are demonstrated, discussed and...
Radiographic Anatomy - Celiac Trunk Angiogram
In this video, Dr. Akram Jaffar, Ph.D., presents a comprehensive overview of the branches of the celiac trunk and their distribution. By watching the video, you will learn to identify important arteries...
Anatomical Basis of Thoracic Outlet Syndrome
Where is the thoracic outlet located? How to differentiate it from the thoracic inlet? What are the anatomical structures that could be compressed as they pass through the outlet? What are the causes...
EMR of Periappendiceal Lesion After Partial Circumferential...
Video case: polyp located close to appendiceal orifice. Minding quite thin wall of the ceacum after proper lifting lateral incision was made to facilitate ensnaring the lesion. Clips applied to the resection...
Featured channels
Mitral Valve Repair Center at The Mount...
As one of the world's most respected and experienced heart…
EUS - ENDO
EUS-ENDO will offer one day of live transmission from Paoli-Calmettes…
SMIT Congress
The idea of this meeting fits excellently into the philosophy…